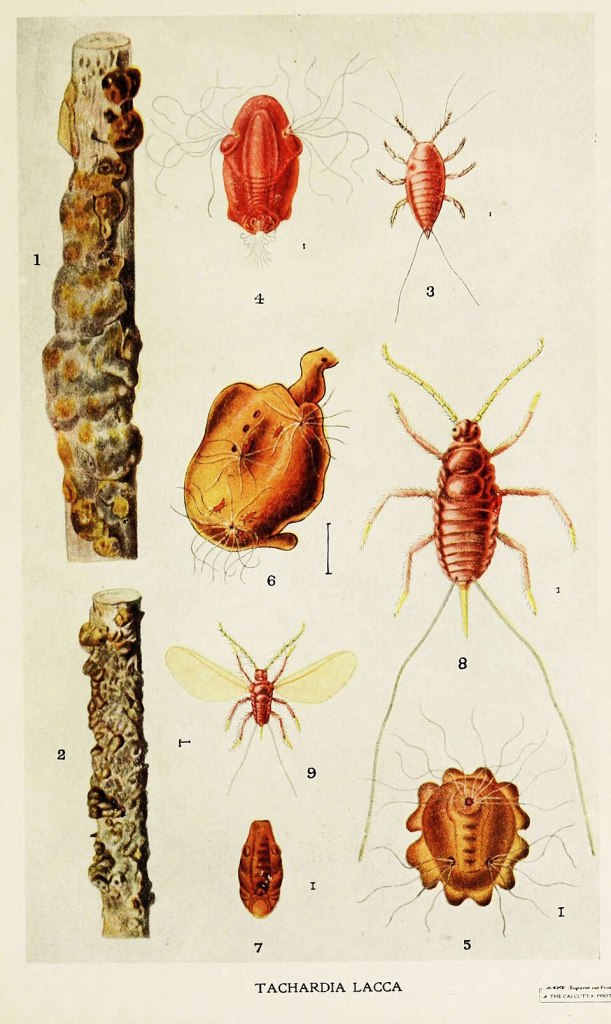
This drawing of the insect Kerria lacca and its lac tubes, by Harold Maxwell-Lefroy, 1909 from flickr, has been made available under the Creative Commons CC0 1.0 Universal Public Domain Dedication.
One of the most colourful associations involving invertebrates and plants is that between the cochineal and the prickly pear cactus*. The cochineal (Dactylopius coccus) is an insect that feeds on Opuntia species (commonly known as prickly pear cactus) and is the source of a brilliant red dye – ‘carmine’ – that’s also known as cochineal. In days gone by the colour was highly prized by people, e.g., for dying the robes of cardinals in the Roman Catholic church, and the uniforms of soldiers known as ‘red-coats’. And “The scarlet dye even found its way back across the ocean, into the “broad stripes” of the embattled banner over Fort McHenry that inspired the U.S. national anthem” (Amy Butler Greenfield). The vibrant red of carmine was also used in art (Elene Phipps), to colour such items as the dress of the ‘Lily Madonna’ [also known as the ‘Virgin and the child’, and – somewhat botanically bizarrely – the ‘Madonna with the iris’] (Marjorie E Wieseman)] by Albrecht Dürer (Ayça Alper Akçay, 2024).
But, the cochineal isn’t the only invertebrate source of a red pigment. Another that might have you seeing red is the lac insect (Kerria lacca). This creature is the source of ‘lac’, “A deep red colorant extracted from the crude shellac resin excreted by the lac insect” [quoted from here]. Like cochineal, lac has been much sought after throughout human history (Pankaj Dagur & Manik Ghosh, 2025), e.g., for dying silk and leather.
The phytophagous (Samantha Murray) lac insect feeds on the sugar-rich sap it extracts from the phloem (Larry Hodgson) of the hosting plant (Ayashaa Ahmad et al., 2012)). The importance of which relationship is underlined in this bold assertion, “Lac insects would not exist without the support of host plants” (Nawaz Haider Bashir et al., 2022). But, unlike the close association of cochineal with prickly pear cacti, the lac insect is associated with a wide variety of host plants (e.g., Kusum (Schleichera oleosa), Palas (Butea monosperma), and Ber (Ziziphus mauritiana) in India (Kewal Krishan Sharma, Rajkumar Pawar) – which varies within the widely-geographically-dispersed countries in which the invertebrate is cultivated.
However, although carmine is apparently made by the cactus-eating cochineal – from the carminic acid the insect produces as a herbivore-deterrent (Sydnee Craven, Katherine Gallagher), the true origin of the lac insect’s colourant has long been a mystery. But, before you think how clever the lac insect is and thank it for all of its hard work in producing the pigment, you need to take a look at the work of Sourajyoti Pal Vaishally et al. (2025).
In trying to solve the puzzle of lac’s ultimate origin, Vaishally et al. (2025) took a deep-dive into the genome of the lac insect. But, they didn’t just examine the insect’s DNA, they also looked into the DNA of two organisms, symbionts, that live inside the insect [i.e., they are therefore endosymbionts], the bacterium Wolbachia (Rupinder Kaur et al., 2021; Jillian Porter & William Sullivan, 2023) and a ‘yeast-like symbiont’ (YLS)**.
Examining the genome of the insect and the bacterium in detail, Vaishally et al. (2025) found no evidence that those organisms had the genetic capacity to synthesise laccaic acid, the common name for the red pigment. They also concluded that the laccaic acid did not come from the plant upon whose phloem sap the insect feeds. However, Vaishally et al. (2025) did discover that the YLS is the sole source of the amino acid (Michael K Reddy) tyrosine that’s a prominent component of the pigment molecule. Furthermore, the YLS “carried the entire set of genes needed for laccaic acid production” (Linda Stewart)***. Which explains the scientific article’s succinct and informative title, “An endosymbiotic origin of the crimson pigment from the lac insect”.
Further support of involvement of the YLS in this chemical synthesis came from the use of fungicide. When treated with this fungus-killing compound, insects were reduced in size and weight, and had much-lowered concentrations of laccaic acid (Vaishally et al., 2025)****.
The value of this work is stated by the final sentence of the scientific paper’s Discussion section, “these findings highlight the role of fungal symbionts in shaping insect adaptation and in synthesizing biologically active and commercially important secondary metabolites such as pigments” (Vaishally et al., 2025).
However, if determining the origin of the lac pigment is one mystery that’s now [probably] solved, another remains: What does the pigment do? Or, in the words of Vaishally et al. (2025), “the biological function of these pigments remains undetermined, they possibly provide defense against predators, or pathogens and parasites that can grow within the vicinity of the resin, or protect against UV damage [references supplied]”. That’s what great about science, in answering one question, there’s always another one to ask.
For more on the lac pigment YLS work, see Linda Stewart, Ranjini Raghunath, Anubha Jain, Asra Mavad, ‘PG’, Chethan Kumar, here, Sneha Khedkar, and here.
* For more about the biology, ecology, and human relevance of cochineal, see Luke Yoquinto; Thomas Eisner et al., 1980; here; Brittney J Miller; Amy Butler Greenfield; here; here; Jireh Nelson; Helen Soteriou & Will Smale; Chris Cooksey, 2018; Rw Dapson, 2007; Devon Van Houten Maldonado; Katherine Gallagher; Joe Schwarcz; Katie Carman; here; here; here.
** For completeness, it should be noted that existence of Wolbachia and the YLS endosymbioints in the lac insect had previously been reported by Amit Vashishtha et al., 2011.
*** Another fungal dimension to the biology of the lac insect is provided by the finding that the genome of the YLS can produce the amino acids and vitamins that are needed by the insect for its growth and development but which the insect cannot acquire from its plant diet. Reduction in this supply when treated with fungicide probably explains the observed lowered size and weight of such insects. This nutritional aspect – in addition to involvement in lac formation (which secretion is believed to have a protective role for the insect (Sujarwo Sujatmoko, 2009) – establishes a benefit that the insect gets from this association. But, what does the fungus get from it?
Within the insect, the YLS “floats around in the insect’s haemolymph (Vincent Brian Wigglesworth) — the equivalent of animal blood”. But, the YLS is also transmitted to the next generation by entering into an oocyte (a developing egg cell) from which it emerges to occupy its new host. Maintenance of the YLS within the insect in this way certainly suggests that its presence there is of some benefit to the fungus. It thus appears that both insect and fungus may benefit from this arrangement, which may therefore represent a mutually-beneficial symbiotic relationship, a mutualism.
**** For consistency of interpretation – that the fungus is the source of the dye – reduction in laccaic acid content is presumably here an indication that the fungicide treatment didn’t kill all of the YLS. One way of answering this question definitively is to see if the YLS can be cultured and grown outside of the insect’s body and produce the dye on its own. Unfortunately, at present, culturing the YLS outside of the insect is not possible (Vaishally et al., 2025).
Interestingly, Rasmus Frandsen et al. (2018) report manufacture of carminic acid [produced by cochineal] using a fungal system that represents an “important step towards industrial-scale production of carminic acid via liquid-state fermentation using a microbial cell factory”. Although this synthetic route will reduce pressure on exploitation of cochineal insects in the environment, quite what the knock-on effects to the economics of traditional cochineal harvesting and the people who are involved in that industry probably remain to be determined.
And, it is also noteworthy that a fungal symbiont has been identified within Kermes quercus (Elżbieta Podsiadło et al., 2018). K. quercus is one of several species in the genus Kermes, oak-tree-sap-sucking insects that are the source of kermes. Kermes is another red pigment (alternatively known as crimson, or kermesic acid), used since antiquity and originally sourced from the Mediterranean region. In light of Vaishally et al. (2025)’s revelation about the lac insect and its YLS, it will be interesting to know what contribution the ‘Kermes fungus’ may make to dye production in that invertebrate.
REFERENCES
Ayashaa Ahmad et al., 2012. Mouthparts and stylet penetration of the lac insect Kerria lacca (Kerr) (Hemiptera: Tachardiidae). Arthropod Structure & Development 41(5): 435-441; https://doi.org/10.1016/j.asd.2012.04.001
Ayça Alper Akçay, 2024. The use of insect pigment in art works. Insects 15(7): 519; doi: 10.3390/insects15070519
Nawaz Haider Bashir et al., 2022. Unraveling the role of lac insects in providing natural industrial products. Insects 13(12): 1117; doi: 10.3390/insects13121117
Chris Cooksey, 2018. The red insect dyes: carminic, kermesic and laccaic acids and their derivatives. Biotechnic & Histochemistry 94(2): 100–107; https://doi.org/10.1080/10520295.2018.1511065
Pankaj Dagur & Manik Ghosh, 2025. Lac dye: Beyond textiles and tradition, a comprehensive review of its bioactive potential. Pharmacognosy Magazine 0(0): doi:10.1177/09731296241305858
Rw Dapson, 2007. The history, chemistry and modes of action of carmine and related dyes. Biotechnic & Histochemistry 82(4–5): 173–187; https://doi.org/10.1080/10520290701704188
Thomas Eisner et al., 1980. Red cochineal dye (carminic acid): Its role in nature. Science 208(4447): 1039-1042; doi: 10.1126/science.208.4447.1039
Rasmus JN Frandsen et al., 2018. Heterologous production of the widely used natural food colorant carminic acid in Aspergillus nidulans. Sci Rep 8: 12853; https://doi.org/10.1038/s41598-018-30816-9
Rupinder Kaur et al., 2021. Living in the endosymbiotic world of Wolbachia: A centennial review. Cell Host & Microbe 29(6): 879-893; https://doi.org/10.1016/j.chom.2021.03.006
Elżbieta Podsiadło et al., 2018. Yeast-like microorganisms in the scale insect Kermes quercus (Insecta, Hemiptera, Coccomorpha: Kermesidae). Newly acquired symbionts? Arthropod Structure & Development 47(1): 56-63; https://doi.org/10.1016/j.asd.2017.11.002
Jillian Porter & William Sullivan, 2023. The cellular lives of Wolbachia. Nat Rev Microbiol 21: 750–766; https://doi.org/10.1038/s41579-023-00918-x
Sujarwo Sujatmoko, 2009. Parasites and predators of Laccifer lacca Kerr on lac culture in East Sumba, East Nussa Tenggara. Journal of Forestry Research 6(2): 119-125.
Sourajyoti Pal Vaishally et al., 2025. An endosymbiotic origin of the crimson pigment from the lac insect. Proc. Natl. Acad. Sci. USA 122(25): e2501623122; https://doi.org/10.1073/pnas.2501623122
Amit Vashishtha et al., 2011. Co-existence, phylogeny and putative role of Wolbachia and yeast-like symbiont (YLS) in Kerria lacca (Kerr). Curr Microbiol. 63(2): 206-212; doi: 10.1007/s00284-011-9961-x

Leave a comment